Most people consider brushing and flossing to be annoying but necessary habits to protect their teeth. A new study led by NYU Shanghai Provost and Distinguished Global Professor of Public Health Bei Wu suggests the stakes may be considerably higher — the community of microorganisms living in your mouth could be linked to how quickly your body ages overall.
The study, recently published in The Journals of Gerontology: Series A: Biological Sciences and Medical Sciences, found that older adults with less diverse oral microbiomes tended to show higher levels of frailty, a medical term for the gradual erosion of the body’s physical reserve. Frailty makes people more vulnerable to illness, hospitalization, and a longer, harder recovery. The findings point to the mouth as an underappreciated window into systemic aging.
The human mouth is home to more than 700 species of microorganisms. This ecosystem, the oral microbiome, plays a role not just in oral health, but in regulating inflammation and supporting broader bodily functions. But when it comes to aging research, it has received far less scientific attention than the gut microbiome, despite its size and complexity.
“The oral microbiome is the body’s second largest microbial community, and it is increasingly recognized as important in inflammation and systemic health,” Wu explained. “We wanted to understand whether what lives in your mouth might actually reflect, and potentially shape, how well your body is holding up as you get older.”
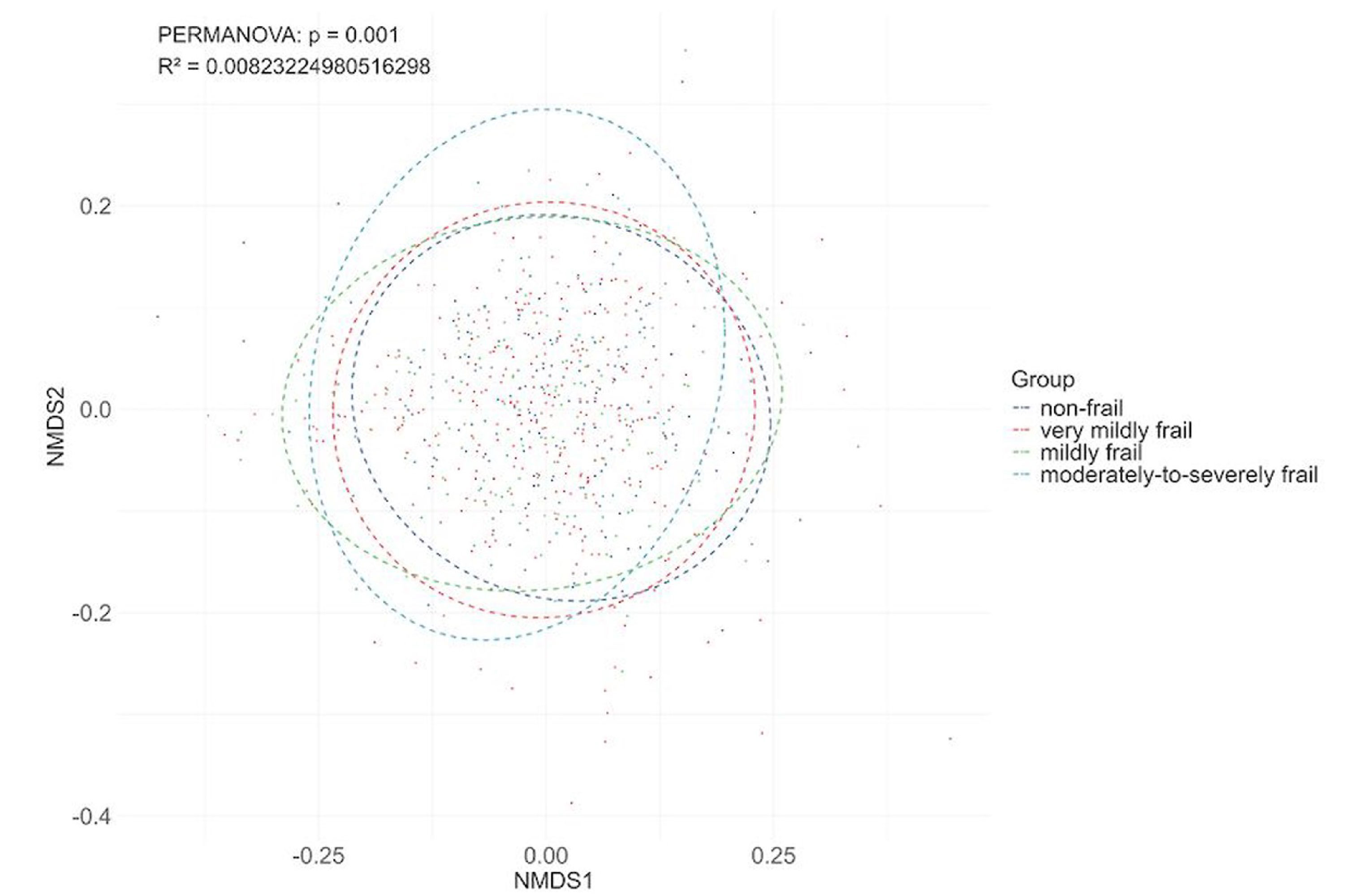
Wu and her colleagues analyzed data from 2,696 adults aged 50 and older drawn from the National Health and Nutrition Examination Survey (NHANES), a nationally representative study in the United States that combines health interviews with laboratory testing. The team measured oral microbial diversity using four established indices and assessed frailty using a 36-item Frailty Index covering cardiovascular, respiratory, musculoskeletal, neurological, and other health domains.
The results were consistent across all four diversity measures: lower oral microbial diversity was associated with higher frailty scores, even after accounting for age, body mass index, education, smoking habits, alcohol use, and other factors. The relationship appeared to be gradual — not a cliff-edge threshold, but a steady pattern in which each step down in microbial richness corresponded to slightly greater frailty burden.
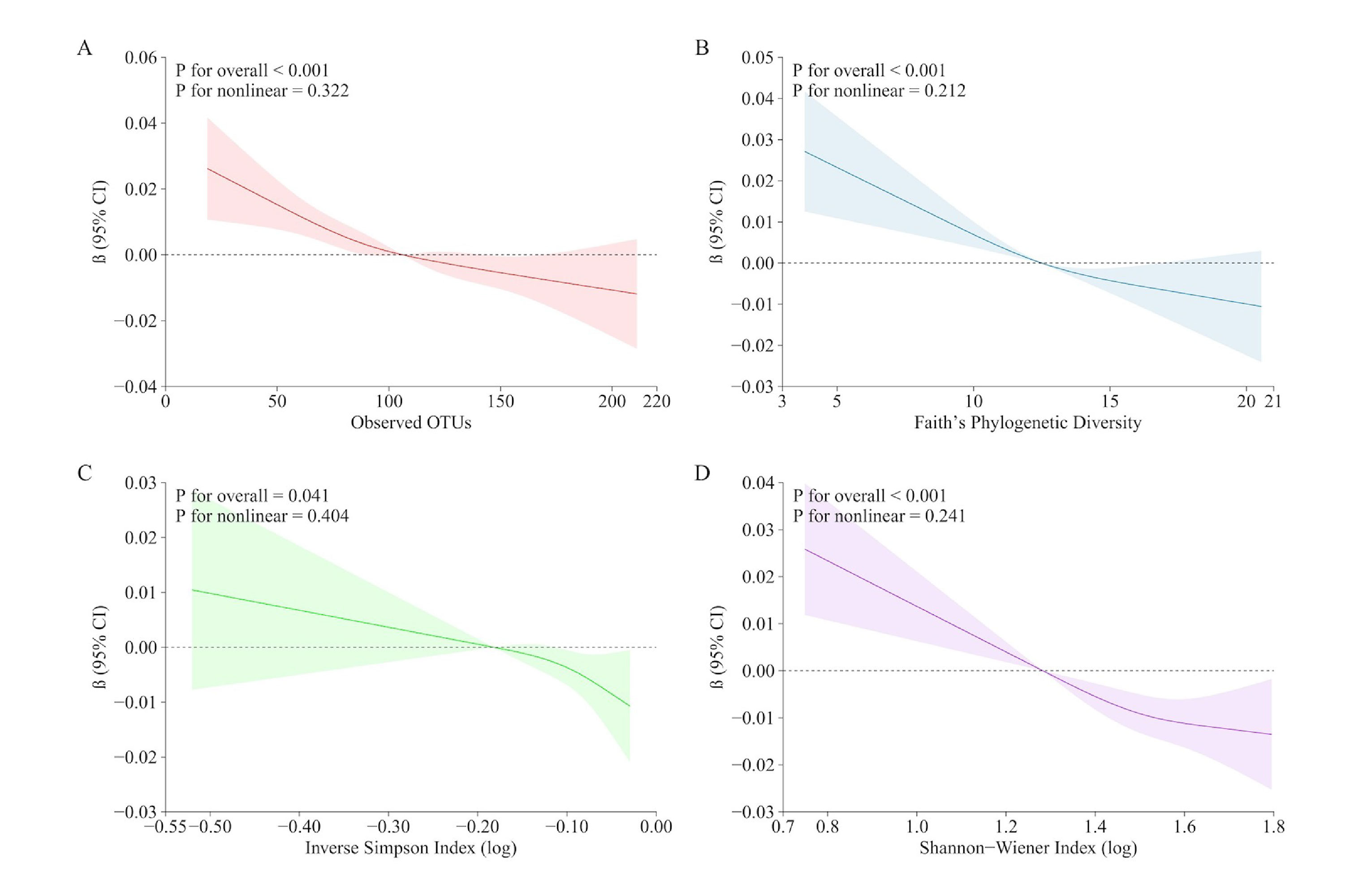
To push the analysis further, the research team applied a method called Mendelian randomization, which uses genetic variants as a kind of natural experiment to test whether a relationship may be causal rather than coincidental.
This genetic analysis identified specific bacterial groups in saliva and on the tongue that appeared to either protect against frailty or heighten its risk. Notably, the same bacterial family could play opposite roles depending on where in the mouth it was found. The results suggest that it is not just the overall diversity of the oral microbiome that matters, but the particular balance of species within it.
“These findings suggest that the relationship is not simply one-way, where poor health leads to a less diverse microbiome,” said first author Ting Xu of Nanjing Medical University. “Some of these specific bacterial taxa may be actively involved in the biological processes that drive frailty. That opens a different kind of conversation about whether we can intervene.”
“The effect sizes we observed, while consistent and statistically significant, are modest,” Xu noted. "Because our population data captures a single point in time rather than tracking people over years, we cannot yet say with certainty how oral microbiome changes and frailty progression unfold together. Longitudinal studies are the important next step.”
Still, the study adds to a growing body of evidence that oral health and whole-body health are deeply interconnected, said Wu. Diet, in particular, appears to play an important upstream role: sugar-heavy diets tend to favor pro-inflammatory bacteria, while plant-based and anti-inflammatory dietary patterns are associated with a more beneficial oral microbial profile.
“The mouth is not an isolated environment,” Wu said. “What happens there can ripple outward in ways we are only beginning to understand. Maintaining good oral health, and the habits that support it, may be one of the more accessible things people can do to support healthier aging.”
The research team includes collaborators from NYU Rory Meyers College of Nursing, NYU Shanghai, Nanjing Medical University, Shenzhen University, and Guangzhou Medical University.